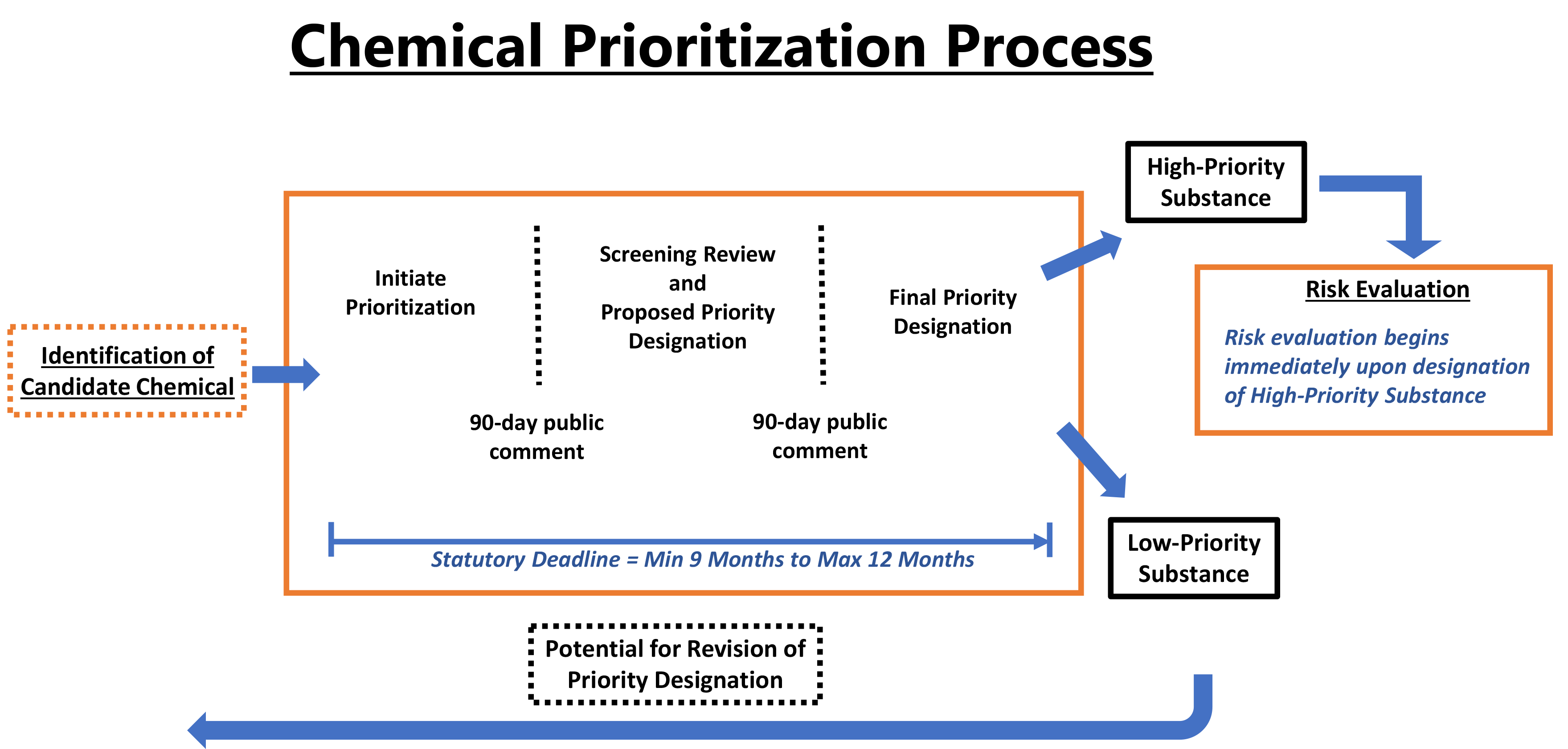
Overview of EPA’s chemical prioritization process (reproduced from EPA’s TSCA website)
Under the amended Toxic Substances Control Act (TSCA), the U.S. EPA plans to initiate prioritization of 40 chemicals from the existing chemical inventory—at least 20 Low-Priority and 20 High-Priority candidates—by the end of calendar year 2018. By December 22, 2019, EPA will have designated at least 20 substances as Low-Priority substances and initiated risk evaluations on at least 20 High-Priority substances. While EPA is currently putting significant effort into developing the process for chemical substance prioritization, this effort is focused primarily on High-Priority determination, with little discussion of how substances will be determined to be Low-Priority.
At their December 2017 public meeting on Approaches for Identifying Potential Candidates for Prioritization for Risk Evaluation, EPA stated that they are seeking input on how to best select and demonstrate Low-Priority. It has been recommended that a starting point may be the Safer Chemical Ingredient List (SCIL) from their Safer Choice Program. However, it is uncertain whether all required TSCA prioritization criteria have been evaluated in developing the SCIL. Also, chemical substances that may benefit from a Low-Priority designation may not be on the SCIL. Manufacturers and processors can contribute to EPA’s process by submitting a case for Low-Prioritization of strategically selected chemicals or groups of chemicals.
Why contribute to the selection of Low-Priority candidates?
There may be a competitive advantage to be derived from being able to tell the market that a chemical or group of chemicals has received an official Low-Priority designation from EPA. Further, receiving a Low-Priority designation may relieve concerns about chemical safety and the possibility of future restrictions being imposed on a chemical or group of chemicals, thus eliminating uncertainties about impacts on investment in those substances.
In these situations, developing a pre-prioritization for a chemical may be a great strategy. The results will provide information to support internal product stewardship decision making, and it may be possible to use the results to facilitate EPA naming the chemical as a Low-Priority candidate and giving it a Low-Priority designation.
How might a pre-prioritization to support a Low-Priority designation be done?
Pre-prioritization to support Low-Priority designation by EPA will involve developing sufficient information to establish that the general standard from TSCA §2605(b)(1)(B)(ii) is met, and that a substance does not present an unreasonable risk of injury to health or the environment through routes of exposure that exist under the conditions of its use. In effect, this says that a Low-Priority substance is one that is not determined to be a High-Priority substance. Other sections of TSCA provide added information about what a Low-Priority substance is not:
- It is not a substance listed in the 2014 Update of the TSCA Work Plan that has a persistence & bioaccumulation score of 3, is a known carcinogen, or has high acute or chronic toxicity
- It is not a substance that is known to or suspected of causing or contributing to cancer, gene mutations, or birth defects (TSCA §2603(e)).
While EPA continues to develop the prioritization approach to be used and is evaluating a number of alternative approaches, there are common considerations that provide further insight as to what may support a Low-Priority designation. The 2014 Update of the TSCA Work Plan and the TSCA Work Plan Chemicals: Methods Document includes a screening process to identify High-Priority candidates. These are indicative of the criteria that may need to be addressed for demonstrating Low-Priority. The screening criteria to be identified as a candidate for High-Priority are:
- Chemicals identified as potentially of concern for children’s health (e.g., chemicals with reproductive or developmental effects)
- Chemicals identified as persistent, bioaccumulative, and toxic (PBT)
- Chemicals identified as probable or known carcinogens
- Chemicals used in children’s products
- Chemicals used in consumer products
- Chemicals detected in biomonitoring programs.
Chemicals that met these screening criteria were then prioritized further by assigning them a high, moderate, or low (3,2, or 1) score, based on:
- Hazard
- Exposure
- Potential for persistence and/or bioaccumulation.
So, for example, if a substance scored low on these criteria, that would support a low prioritization.
TSCA §2605(b)(1)(A) also provides insight by specifying that the process for prioritization of a chemical substance or category of chemical substances[1] shall include consideration of:
- Hazard and exposure potential
- Persistence and bioaccumulation
- Potentially exposed or susceptible subpopulations (such as infants, children, pregnant women, workers, or the elderly)
- Storage near significant sources of drinking water
- Conditions of use or significant changes in the conditions of use of the substance
- Volume or significant changes in the volume manufactured or processed.
ToxStrategies can provide expert assistance in pre-prioritization of chemical substances under TSCA. ToxStrategies has competency with the alternative approaches, models, and supporting databases that are used for chemical substance prioritization. We are experts in the use of high-throughput screening data and in vitro–to–in vivo extrapolation (IVIVE) to predict relative hazard, and high-throughput exposure models to support margin of exposure (MoE) and activity:exposure ratios (AERs). We understand the key factors that chemical substance prioritization must address. We are ready to use these tools to assist chemical manufacturers and processors with pre-prioritization of substances and groups of substances, including to develop submittals to support a Low-Priority designation, based on the considerations and criteria discussed above.
[1] EPA is exploring the organization of chemical substance prioritization by functional use categories as a way to move groups of chemicals through the prioritization process. Examples of functional use categories are adhesion promoters, catalysts, surfactants, and chelators.
